Active smoking has been recognized as a critical lifestyle factor for cardiovascular, respiratory, and neoplastic diseases and contributes to the leading causes of preventable morbidity and mortality [1, 2]. DNA methylation, one of the main forms of epigenetic modification, is involved in the pathways of smoking and smoking-induced diseases [3, 4]. Previous epigenome-wide association studies (EWASs) based on whole blood samples have successfully discovered an increasing number of tobacco smoking-related CpG sites in various genes, such as AHRR and F2RL3 [5, 6, 7]. These DNA methylation patterns have been shown to be useful as quantitative biomarkers to reflect both current and lifetime smoking exposure and to enhance the prediction of smoking-related risks [8, 9, 10, 11].
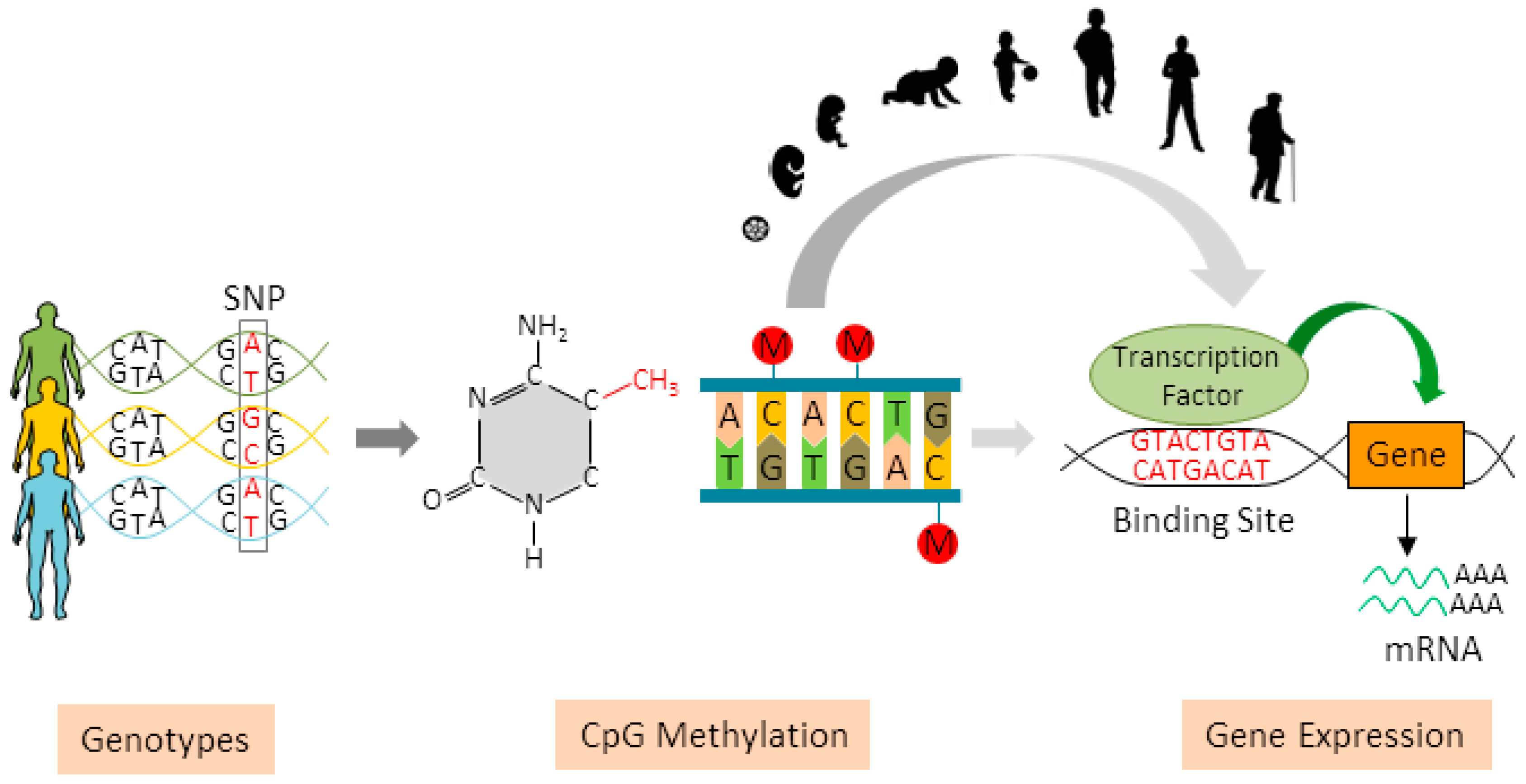
DNA methylation of particular genomic loci might be influenced by neighboring genetic sequence variants [12]. The single nucleotide polymorphisms (SNPs) that are associated with methylation levels of CpG sites are known as methylation quantitative trait loci (mQTLs) [13]. This genetic effect has been determined across different tissues [13, 14, 15, 16] and has been highlighted in several diseases, including neurological disorders, arthritis, and cancer [17, 18, 19, 20, 21]. Recently, the mQTLs have been further reported to play a modifying role in the associations between DNA methylation levels at specific CpG sites and environmental exposures. For instance, Zhang et al. identified 238 mQTLs that were associated with 65 alcohol dependence-related CpG sites in African Americans and 305 mQTLs for 44 unique CpG sites in European Americans [22]. In 2016, Gonseth et al. found out that three of the strongest maternal smoking-related CpG sites in newborns were significantly associated with SNPs located in the vicinity of each gene [23]. Thus, these hereditary traits provide a possible mechanism by which methylation patterns could be different under environmental exposures, if the distribution of risk alleles differs between the exposed and the unexposed. In addition, the linkages of epigenetic signatures to genotypes might also further provide more mechanistic evidence on the genetic and environmental risk factors for various forms of diseases [24].

An integrative study from Heyn et al. Now provides a comprehensive catalog of DNA methylation quantitative trait loci of cancer-risk alleles associated with the most common solid tumor types. The authors demonstrate that DNA methylation linkage analysis is a powerful tool for functional interpretation of genetic risk variants identified in GWAS that otherwise lack mechanistic explanation. Keywords: methylation quantitative trait loci (mQTL), promoter CpGs, alcohol dependence risk genes, genome-wide association, functional annotation, alcohol dehydrogenase (ADH) Introduction DNA methylation usually occurs at CpG sites in somatic cells, and is non-randomly distributed across the genome.
However, such genetic influences have not been well addressed or even overlooked by previous EWASs of active smoking exposure; to our knowledge, no study has so far investigated their contributions to the methylation intensities of active smoking-related CpG sites and smoking-related health outcomes in the general population. Therefore, we conducted a comprehensive analysis in a large population-based study of older adults in Germany with the aim of exploring the hitherto unknown association between active smoking-related DNA methylation and individual genetic variations. In particular, we aimed to identify the mQTLs within ± 50 kb from each of 151 previously reported active smoking-related CpG sites in whole blood samples [25] and to assess their relationships with active smoking exposure and all-cause mortality.
